Apoptotic Bodies (ApoBDs) and ApoMVs (Up to 5 μm)
• Biogenesis: Released during the "controlled demolition" of a cell during programmed cell death. They are
unique for containing intact organelles and chromatin fragments. The formation process involves three
main steps: membrane vesicle formation, apoptotic membrane protrusion, and division into apoptotic
bodies. Actin contraction mediates repeated cycles of membrane blebbing and retraction. ROCK1 is a key
regulatory kinase, while Pannexin 1 (PANX1) channels serve as negative regulators of apoptotic cell
catabolism. Smaller apoptotic vesicles (ApoEVs, 0.1–1 μm) are also produced during this process.
• Discovery: Formally described by Kerr, Wyllie, and Currie in 1972. They used light and electron
microscopy to define the morphological sequence of apoptosis: chromatin condensation, membrane
blebbing, and disintegration into membrane-bound vesicles.
• Key Markers: Annexin V (binds PS), Histones, DNA fragments, Thrombospondin.
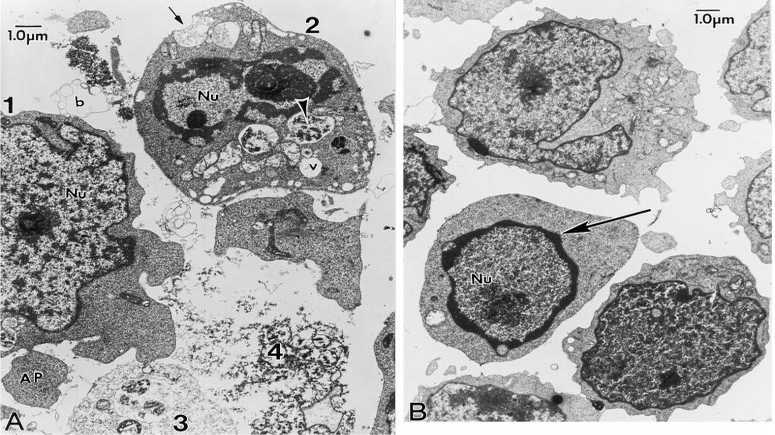
The various phases of apoptotic body formation in chemotherapeutic agenttreated ALL-697 cells. (A) ALL-697 cells were treated with 10 M etoposide for 6 h. Various phases of the formation of apoptotic bodies are evident in this micrograph and are labeled 1-4. The cell labeled 1 is at an early stage and shows very minimal chromatin condensation in the nucleus (Nu), but an apoptotic body (AP) can be seen budding off and another is probably forming adjacent to it. The cell labeled 2 is at an intermediate stage and shows partially condensed chromatin in the nucleus, apoptotic body formation and many heterogeneous vesicles (V). The black and white arrowhead points to a vesicle with dense contents that suggests lysosomal origin. The small arrow points to one vesicle that appears poised to open and release to the extracellular space, which is full of membrane profiles and debris, characteristic of cells exposed to apoptotic inducers. The cells labeled 3 and 4 have completed apoptosis and are dispersing as granular debris (the ghost cell stage). This images clearly demonstrates that induction of apoptosis is asynchronous. (B) The typical morphological changes of the cells after 3 h treatment with 10 M etoposide. The arrow indicates the beginning of chromatin condensation at the periphery of the nucleus. Of note is that at the early phase of apoptotic body formation, there is not much accumulation of vesicles in the cells
https://www.researchgate.net/figure/The-various-phases-of-apoptotic-body-formation-in-chemotherapeutic-agenttreated-ALL-697_fig5_13109501
Exosomes (30–150 nm): The Endosomal Pathway
• Biogenesis: Formed by the inward budding of late endosomes to create multivesicular bodies (MVBs).
The plasma membrane invaginates to form early sorting endosomes (ESEs), which mature and fuse to
become late sorting endosomes (LSEs). LSEs undergo a second invagination to produce MVBs containing
intraluminal vesicles (ILVs). This process is primarily governed by the ESCRT (Endosomal Sorting
Complex Required for Transport) machinery. Non-classical, ESCRT-independent pathways also exist,
such as Rab31 GTPase-mediated biogenesis. MVBs can either fuse with lysosomes for degradation or
with the plasma membrane to release ILVs as exosomes.
• Discovery: Coined by Dr. Rose Johnstone in 1987 at McGill University. While investigating reticulocyte
maturation, her team used electron microscopy (EM) and immunogold labeling to observe the "reverse
endocytosis" of transferrin receptors, identifying these vesicles as a sophisticated waste-clearance and
communication mechanism. Earlier foundational work includes Harding, Heuser, and Stahl (1983), who
characterized receptor-mediated endocytosis and transferrin receptor recycling in rat reticulocytes.
• Key Markers: Tetraspanins (CD63, CD9, CD81), Alix, TSG101, Flotillin-1.
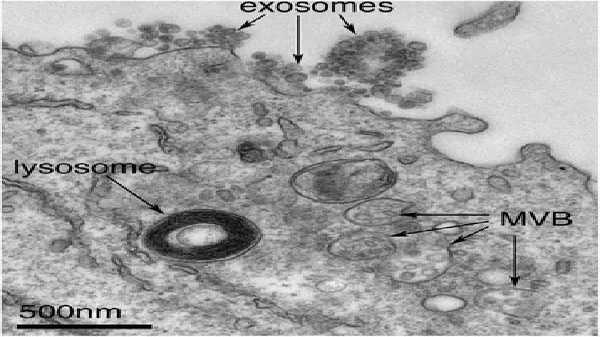
Exosomes correspond to intraluminal vesicles of multivesicular bodies. A transmission electron micrograph of an Epstein–Barr virus-transformed B cell displaying newly expelled exosomes at the plasma membrane. Multivesicular bodies (MVB) can be seen which can deliver content to lysosomes for degradation or can fuse with the cell surface to release intraluminal vesicles as exosomes, indicated by the arrows at the top of the picture
Edgar, J.R. Q&A: What are exosomes, exactly?. BMC Biol 14, 46 (2016). https://doi.org/10.1186/s12915-016-0268-z
Microvesicles / Ectosomes (50–2000 nm): Plasma Membrane Budding
• Biogenesis: Unlike exosomes, microvesicles arise from the direct outward budding and fission of the
plasma membrane. This is driven by phospholipid asymmetry (specifically the translocation of
phosphatidylserine to the outer leaflet) and actomyosin-mediated cytoskeletal contraction. Ca 2+ -dependent
cytoskeletal remodeling plays a key role: elevated calcium activates calpain and scramblase enzymes,
driving membrane asymmetry and budding. Some MVs bud directly from the cell membrane mediated by
ARRDC1 and TSG101 proteins. Rho small GTPases (particularly CDC42) and Rho-associated coiled-coil
containing kinases (ROCK) are essential regulators, with CDC42 serving as a convergence point for
various regulatory signals in MV generation.
• Discovery: Identified throughout the 1990s (notably by Stein, Luzio, and Fox), these were initially
characterized as "procoagulant dust" or "ectosomes" released during cellular activation or
complement-mediated stress. Historical roots trace back to the 1940s, when Chargaff and West studied
blood clotting factors in New York and observed sub-cellular procoagulant particles in platelet-free plasma.
• Key Markers: Integrins, Selectins, CD40, Phosphatidylserine (PS).
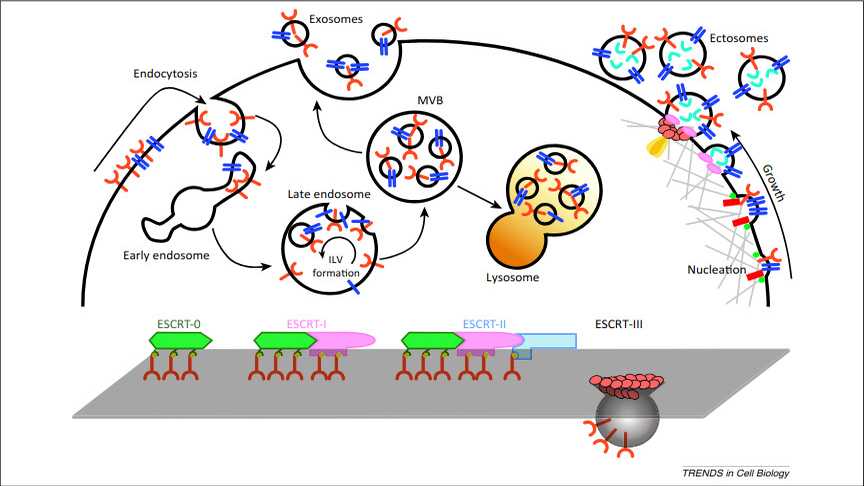
Exosome and ectosome biogenesis. Exosome biogenesis and intracellular life are depicted on the left. Transmembrane proteins (red) are endocytosed and trafficked to early endosomes. Once sorted to late endosomes, the endosomal sorting complex required for transport (ESCRT)-0 complex recruits ubiquitinated proteins, while ESCRT-I and -II mediate the budding of intraluminal vesicles (ILVs). The multivesicular body (MVB) can either follow a degradation pathway fusing with lysosomes (orange) or proceed to release the ILVs as exosomes to the extracellular space by an exocytic step. A summary of the ESCRT cycle as determined from exosome studies is shown at the bottom. Ectosome assembly is illustrated on the right. During nucleation at the plasma membrane, transmembrane proteins (blue) are clustered in discrete membrane domains that promote outward membrane budding. Tetraspanins and other proteins abundant at the domain may have a role by promoting the sorting of other components. Lipidic anchors (myristoylation, palmitoylation) of proteins (green) accumulate proteins in the lumen as well as contributing to membrane curvature. Additional mechanisms of ectosome formation include the Ca2+ -activated scramblases (red rectangles), which randomize the distribution of lipids between the two faces of the plasma membrane. The cytoskeleton (light gray) becomes looser, while cytosolic proteins and RNA molecules (aquamarine) are sorted to ectosomes. Recruited TSG101, a member of the ESCRT-I complex, mediates mobilization to the plasma membrane of ESCRT-III (salmon subunit), which promotes the assembly of a spiral. The specific ATPase VPS4 (orange) mediates the disassembly of the spiral by pulling its end
https://gene-quantification.com/cocucci-meldolesi-exosomes-ectosomes-2015.pdf
Migrasomes: The 2015 "Pomegranate" Breakthrough
Migrasomes represent a distinct class of large organelles (up to 3 μm) that form on retraction fibers at the rear
of migrating cells. They are membrane structures produced at the tips or intersections of contractile filaments
generated during cell migration. As the cell continues to migrate, the contractile filaments break and the
vesicles are released into the extracellular space or phagocytosed by neighboring cells. The process by which
cells release their contents via migrasomes is termed migracytosis.
• Discovery: Reported in 2015 by Li Yu's team (Ma et al.) at Tsinghua University. Using scanning electron
microscopy (SEM) and Tspan4-GFP labeling, they identified these "pomegranate-like" structures
containing multiple intraluminal vesicles.
• Life Cycle:
1. Nucleation: Assembly of SMS2 (sphingomyelin synthase 2) foci at the cell front.
2. Maturation: Recruitment of integrins via the PI(4,5)P 2 and Rab35 axis to anchor the site to the
extracellular matrix.
3. Expansion: Membrane tension fluctuations cause swelling, stabilized by Tetraspanin-4 (Tspan4) and
cholesterol macrodomains.
• Physiological Role: They act as signaling hubs for chemokines (e.g., Cxcl12) and coordinate processes
like angiogenesis, vascular homeostasis, immune response, tissue regeneration, and tumor metastasis.
Notably, they facilitate mitocytosis—the expulsion of dysfunctional mitochondria to maintain cellular
homeostasis.
• Key Markers: Tspan4, SMS2, Integrin α5, Cholesterol.

Migrasomes from RAW264.7 cells. Transmission electron microscopy image of migrasomes after 24 h of cell culture (the left side), Scale bar = 1 μm. The larger magnification image of the white box in the left (the right side), Scale bar = 1 μm. The white arrows indicate the migrasomes
Cell Commun Signal . 2023 May 8;21(1):105. doi: 10.1186/s12964-023-01121-4. Migrasomes, a new mode of intercellular communication
Additional EV Subtypes: Oncosomes and Exophers
Large Oncosomes (LOs) and Cancer Multiomics
Aggressive, amoeboid cancer cells release atypically large EVs (>1 μm) known as large oncosomes. These
double-layer membrane structures can transport proteins (including oncoproteins), cytokines, enzymes, and
lipids, enabling cancer cells to regulate target cell phenotypes and promote tumor metastasis.
• Discovery: Characterized by Dolores Di Vizio's team (2009–2012) using High-Definition Single Cell
Assays (HDSCA) to distinguish them from standard exosomes in prostate cancer patients.
• Mitochondrial Enrichment: Multiomic analysis (mass spectrometry and single-EV RNA-seq) reveals that
LOs are significantly enriched with mitochondrial proteins (e.g., TOMM40, ATP5B) and transcripts
compared to smaller EVs.
• Clinical Utility: LO protein profiles provide a "liquid biopsy" for assessing castration resistance in prostate
cancer. Research in pancreatic cancer suggests that surgical manipulation increases total analytes but
does not necessarily increase circulating tumor cells (CTCs).
• Key Markers: CK18, ARF6, Mitochondrial proteins (TOMM40, ATP5B).Exophers: A Newly Recognized Disposal Mechanism
Exophers are exceptionally large membrane vesicles (~4 μm) first described in Caenorhabditis elegans
neurons. These vesicles are filled with aggregation-prone proteins and damaged organelles, including
mitochondria. Nematode nerve cells use exophers to eliminate harmful intracellular substances. A similar
mechanism has been found in mammalian cardiomyocytes, which excrete dysfunctional mitochondria via
exophers that are subsequently phagocytosed by heart-resident macrophages, thereby maintaining myocardial
health. Research into whether exopher-like pathways exist in other tissues, including the brain, is ongoing.
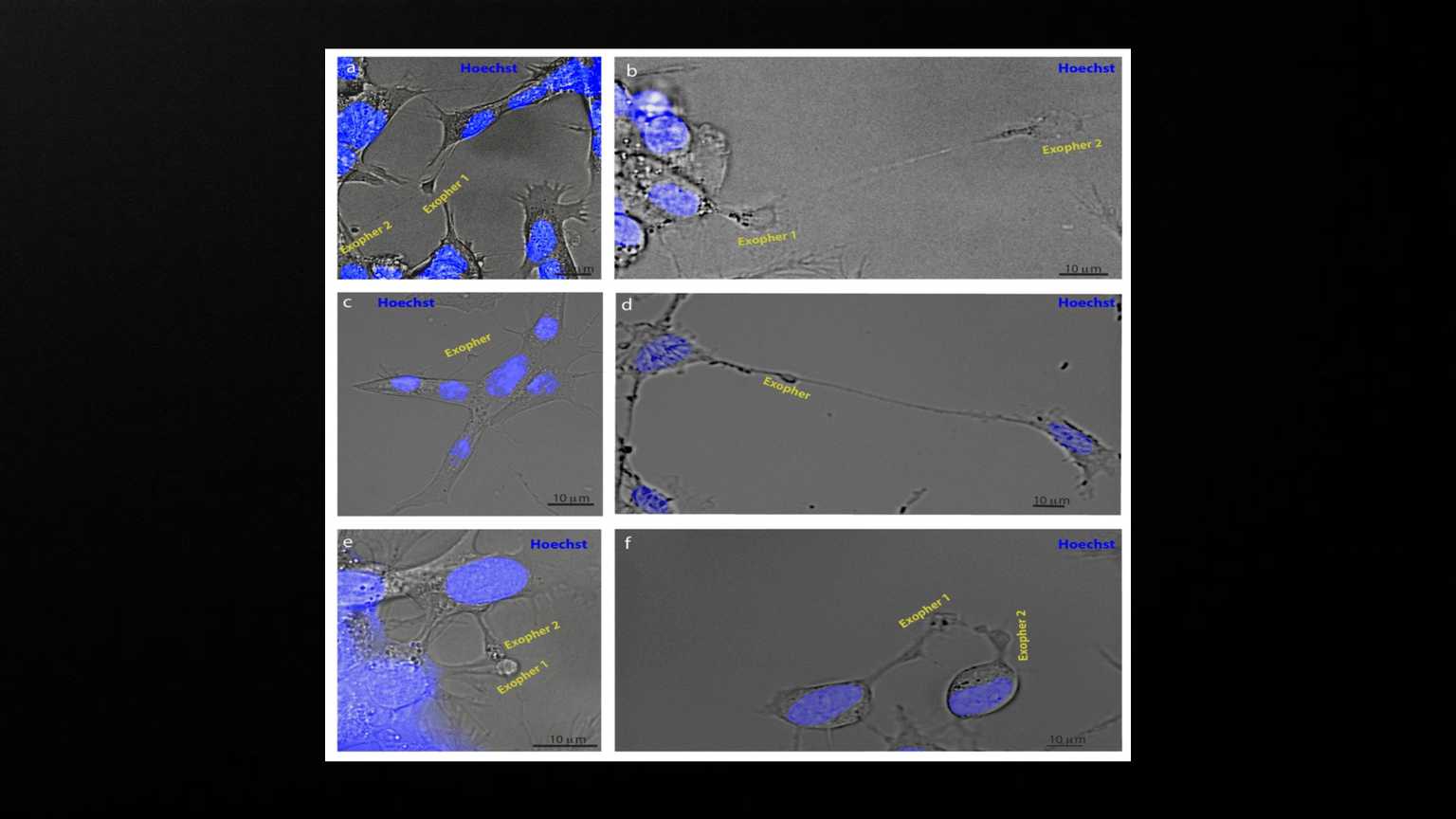
Exophers appear to mediate neuronal communication. SH-SY5Y cells were stained with Hoechst and imaged using brightfield and fluorescence microscopy. a, b) Examples of exophers emanating from another exopher (N = 4 observations). c, d) Examples of exophers connected to two or more different cells via nanotubes (N = 6). e, f) Examples of exophers originating in two adjacent cells in contact with each other (N = 3).
Exophers are components of mammalian cell neurobiology in health and disease December 2021 DOI:10.1101/2021.12.06.471479 ; https://www.researchgate.net/publication/356850720_Exophers_are_components_of_mammalian_cell_neurobiology_in_health_and_disease
